"Andexxa's rapid reversal of the anticoagulating effects of rivaroxaban and apixaban will help clinicians treat life-threatening bleeds, where every minute counts. Join Now to View Patient Handouts Create a FREE Epocrates Online account to access patient. 
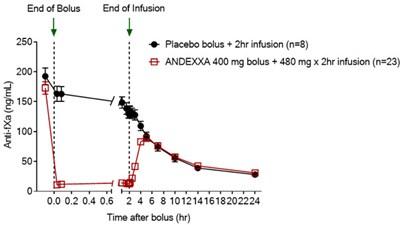
The FDA approval of Andexxa was based on data from two Phase III ANNEXA. This information is currently not available for this drug. "Today's approval represents a significant step forward in patient care and one that the medical community has been eagerly anticipating," ANNEXA-4 chair Stuart J Connolly, MD, McMaster University, Hamilton, Ontario, Canada, said in the release. As a result, the native Factor Xa is available to participate in the coagulation. Follow the same procedure outlined above for IV bolus preparation. Discard the vials, including any unused portion. The median decrease in anti-factor Xa activity from baseline was 90% for rivaroxaban and 93% for apixaban. Transfer the ANDEXXA solution from the syringe into an empty polyolefin or polyvinyl chloride IV bag with a volume of 250 mL or less. Among the 185 evaluable high-risk patients in the open-label study, the agent was shown to provide effective clinical hemostasis in 83% of patients out to 12 hours, as recently reported by | Medscape Cardiology. Additional important safety information and full prescribing information are available here.Īpproval of the agent was supported by data from two phase 3 ANNEXA studies, ANNEXA-R and ANNEXA-A, which demonstrated a median decrease in anti-factor Xa activity from baseline of 97% for rivaroxaban and 92% for apixaban, according to a company news release, posted May 3.Īs part of its review, the FDA also assessed interim data from the ongoing ANNEXA-4 study.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
